Mitochondrial Neuropathy
Leber’s hereditary optic neuropathy (LHON) is an inherited retinal degenerative disorder caused by mitochondrial DNA mutations that lead to retinal cell dysfunction, progressive loss of visual acuity, and eventual vision loss. Mitochondrial dysfunction and oxidative stress are also implicated more broadly in glaucoma and other optic neuropathies, making this a clinically relevant therapeutic target area.
Model Overview
The rotenone-induced mitochondrial neuropathy mouse model is a toxin-based model used to study neurodegeneration driven by mitochondrial dysfunction, with particular relevance to optic neuropathies.
Rotenone is a potent inhibitor of mitochondrial complex I. Following intravitreal administration, it disrupts oxidative phosphorylation, leading to impaired ATP production and increased generation of reactive oxygen species (ROS). This mitochondrial stress preferentially affects retinal ganglion cells due to their high energy demand.
The result is progressive, dose-dependent retinal ganglion cell degeneration, optic nerve damage, and functional deficits. This model recapitulates features of mitochondrial optic neuropathies and is well-suited for evaluating neuroprotective and antioxidant therapeutic strategies, including those relevant to LHON.
Typical Endpoints
- Retinal flatmounts (RGC survival)
- Optic nerve histology (axon counts)
- OCT imaging (ganglion cell complex and total retina thickness)
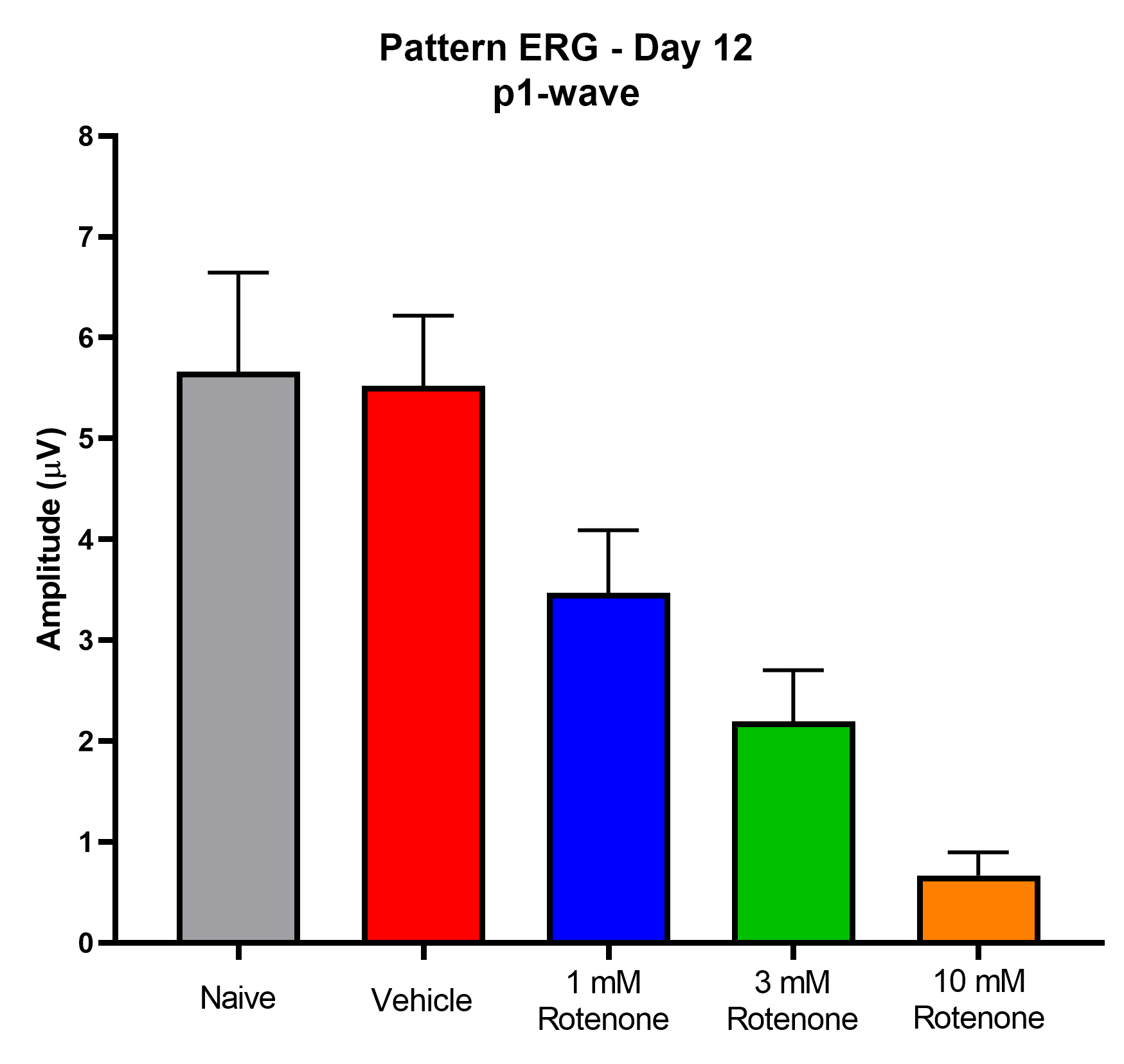
- Pattern ERG (RGC function)
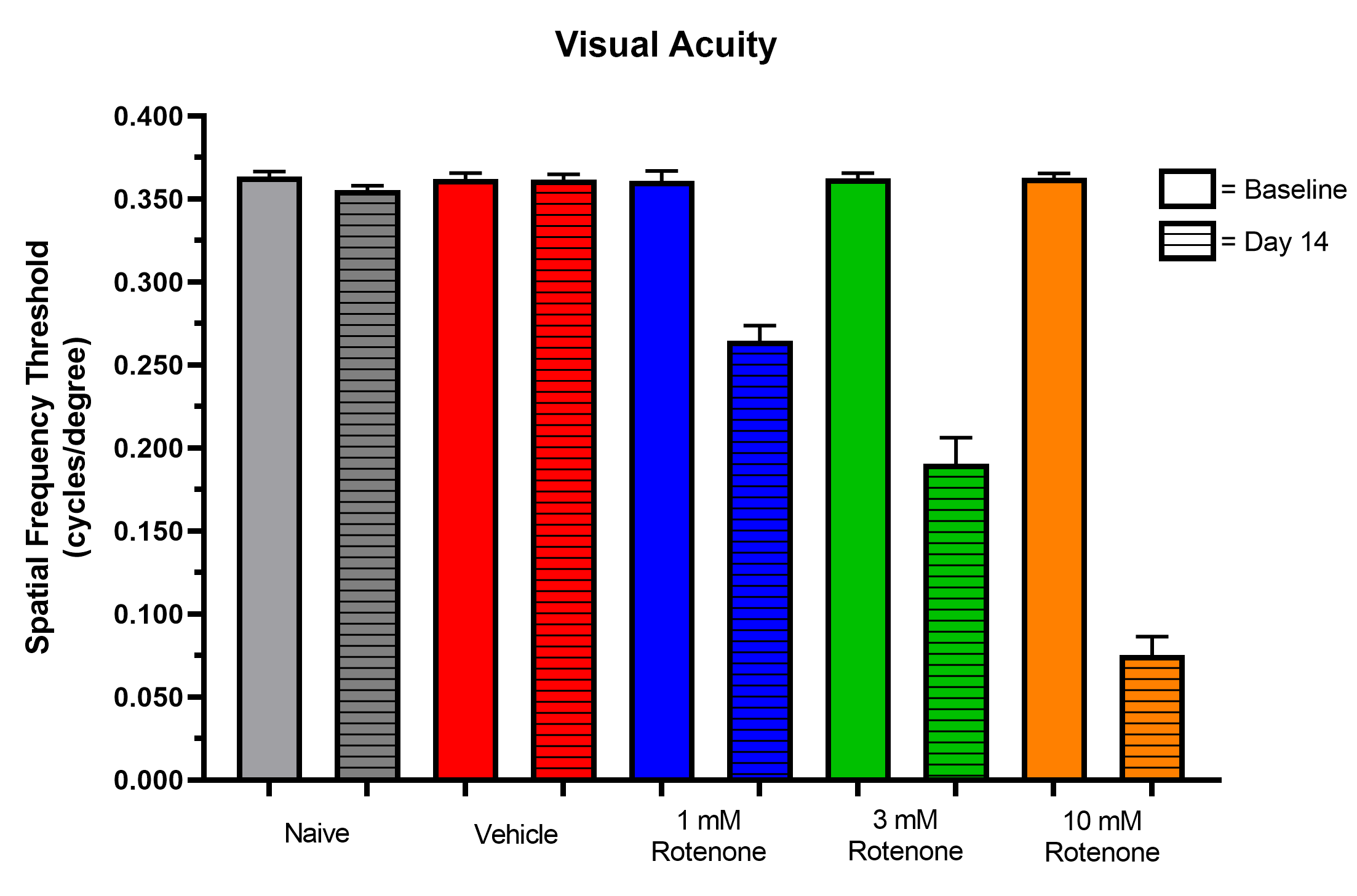
- Optokinetic tracking (visual acuity)
- Retinal histology (cell subtype detection)
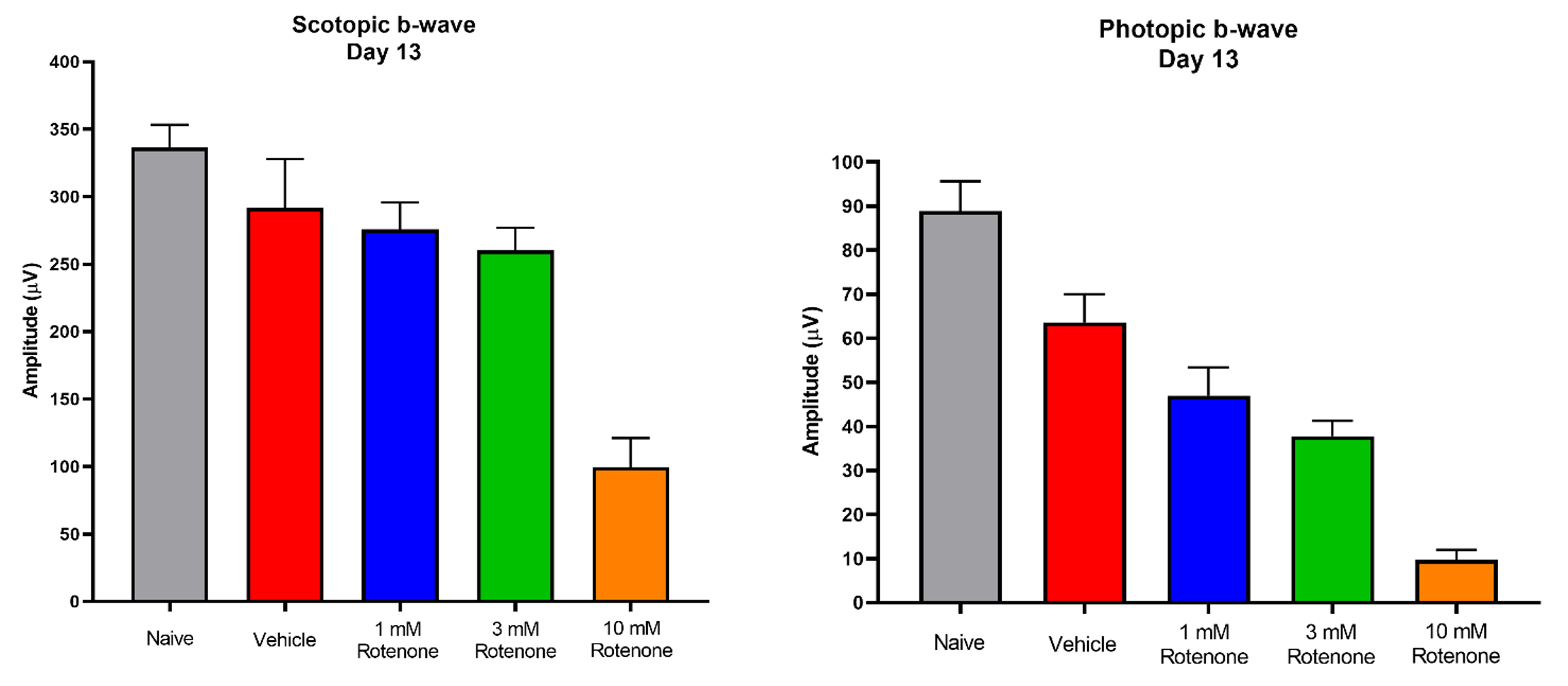
Representative Data
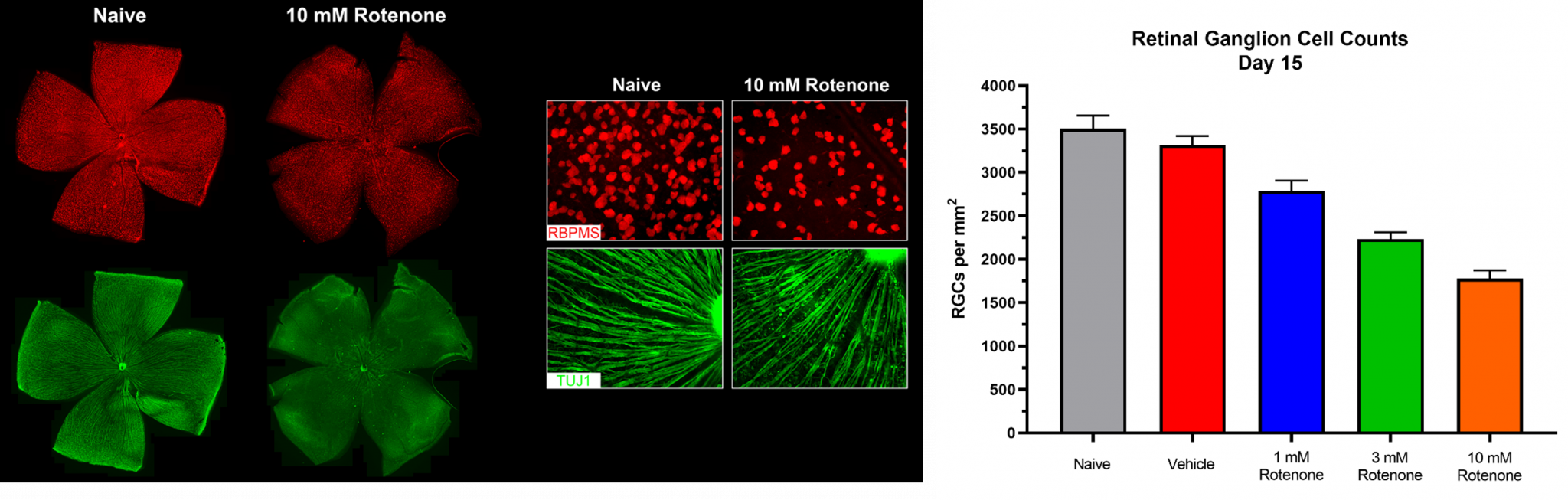
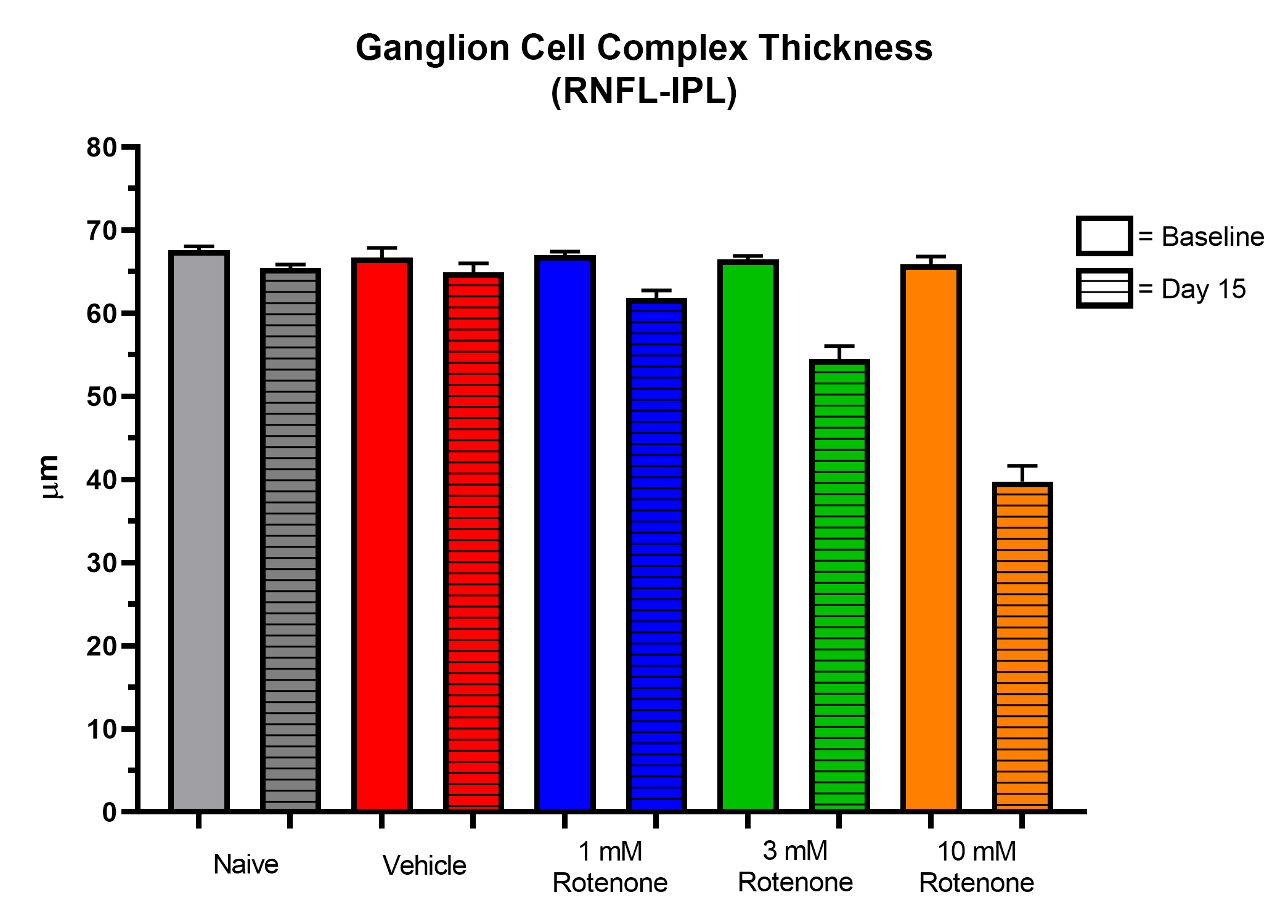
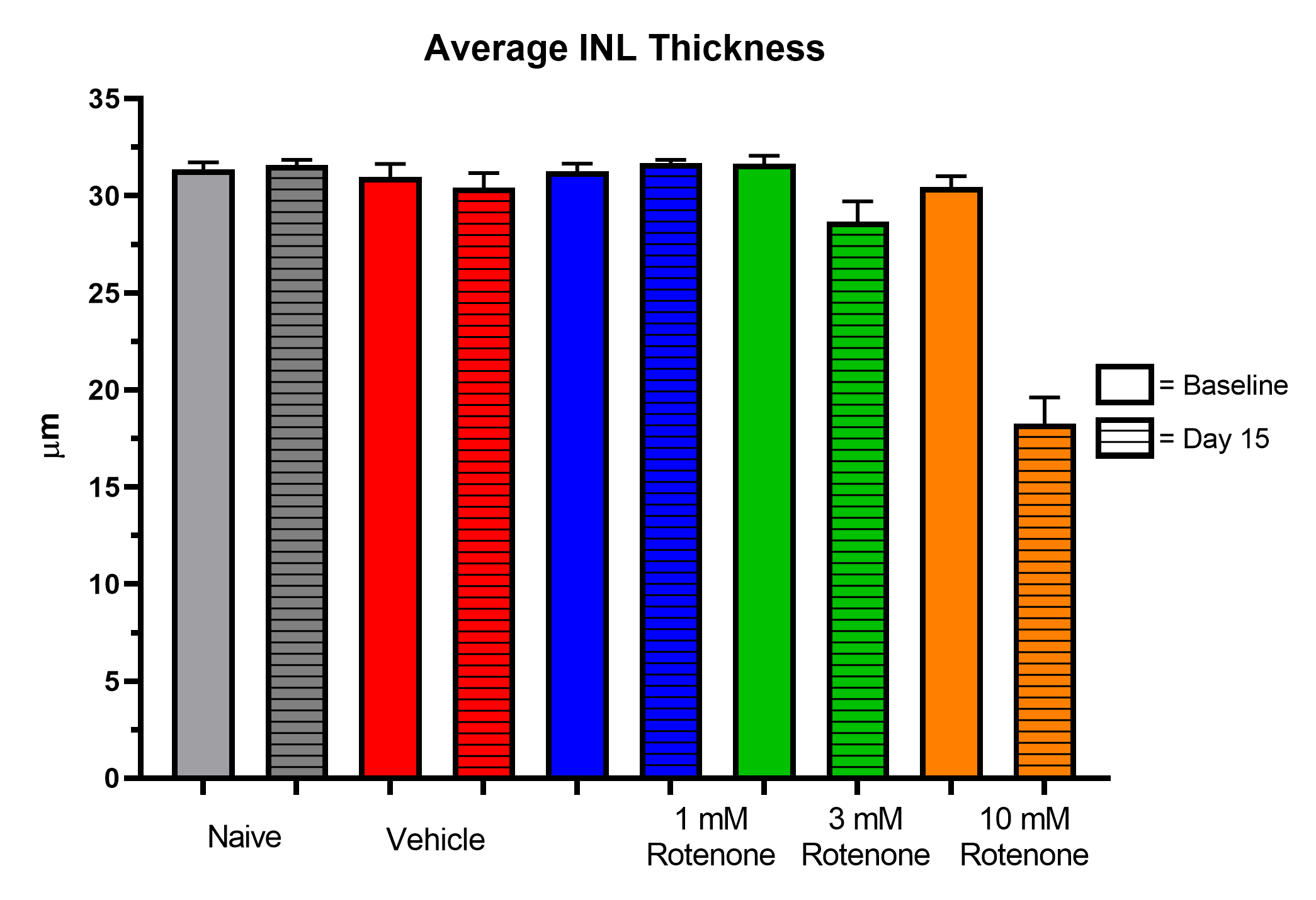
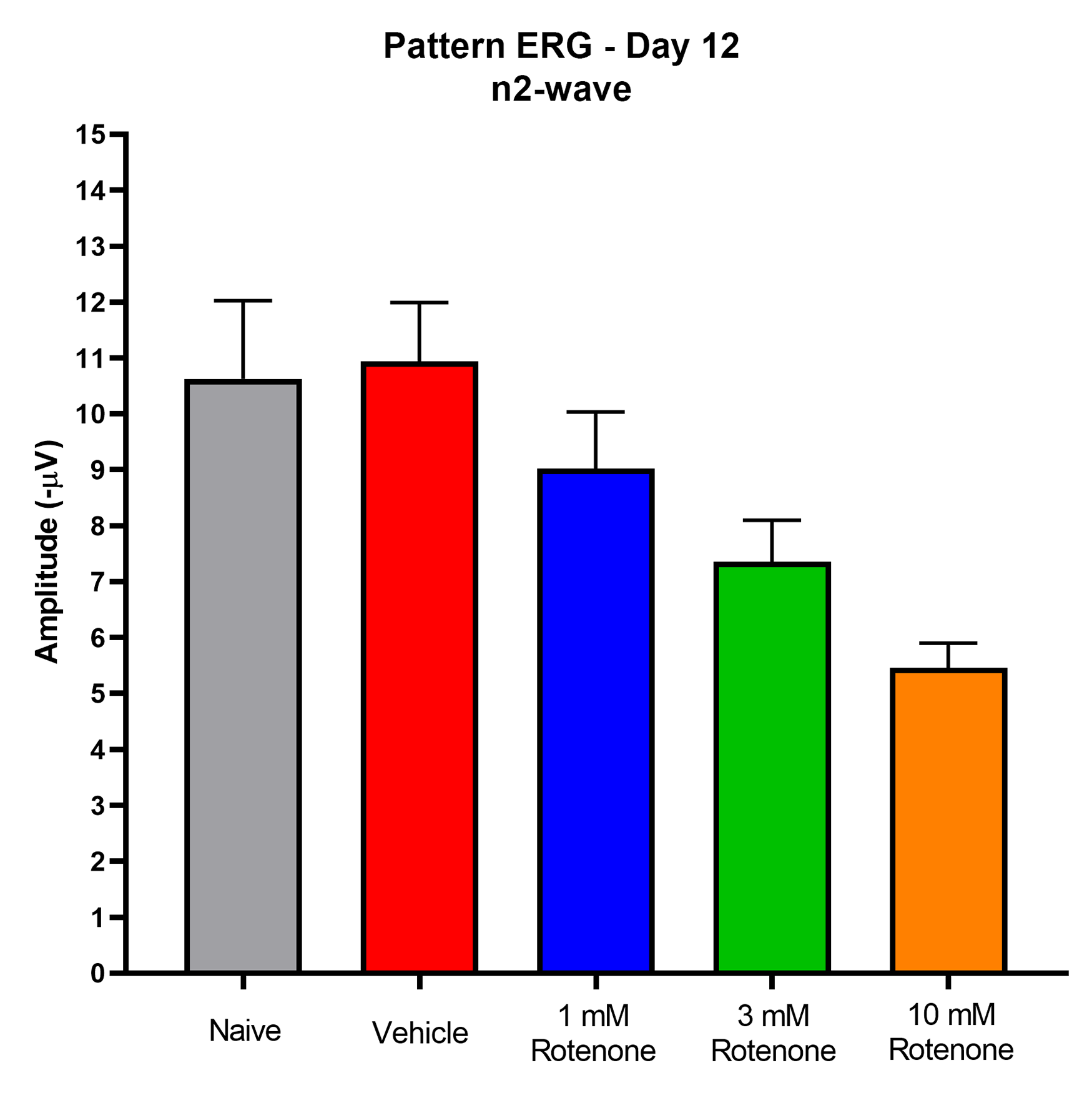
Interested in this model?
We are happy to share more about study design options, endpoints, or timelines.
Contact us to discuss your study goals.

