Optic Nerve Crush
Glaucoma is a group of neurodegenerative optic neuropathies characterized by retinal ganglion cell (RGC) death and optic nerve degeneration. Without—and often despite—current therapeutic intervention, glaucoma ultimately leads to progressive and irreversible vision loss. The initial insult in glaucoma pathogenesis is believed to occur at the lamina cribrosa, a collagenous structure where RGC axons exit the eye and coalesce to form the optic nerve. Injury to the RGC axons triggers a functional decline and progressive loss of RGC somas in the inner retina.
Model Overview
The rodent optic nerve crush (ONC) model involves creating an acute, mechanical injury to the optic nerve at the glial lamina (analogous to the lamina cribrosa in humans) and is widely used to study glaucomatous pathophysiology and evaluate novel therapeutic strategies.
ONC is performed on anesthetized mice or rats by exposing the optic nerve through the bulbar conjunctiva and briefly compressing the nerve just behind the globe using self-closing forceps. Apoptotic RGC death is evident by three days post-crush, followed by Wallerian degeneration of distal axons. Functional deficits can be assessed longitudinally via optokinetic tracking and pattern electrophysiology. This model is highly reproducible and well-suited for evaluating the efficacy of neuroprotective and neuroregenerative therapies.
Typical Endpoints
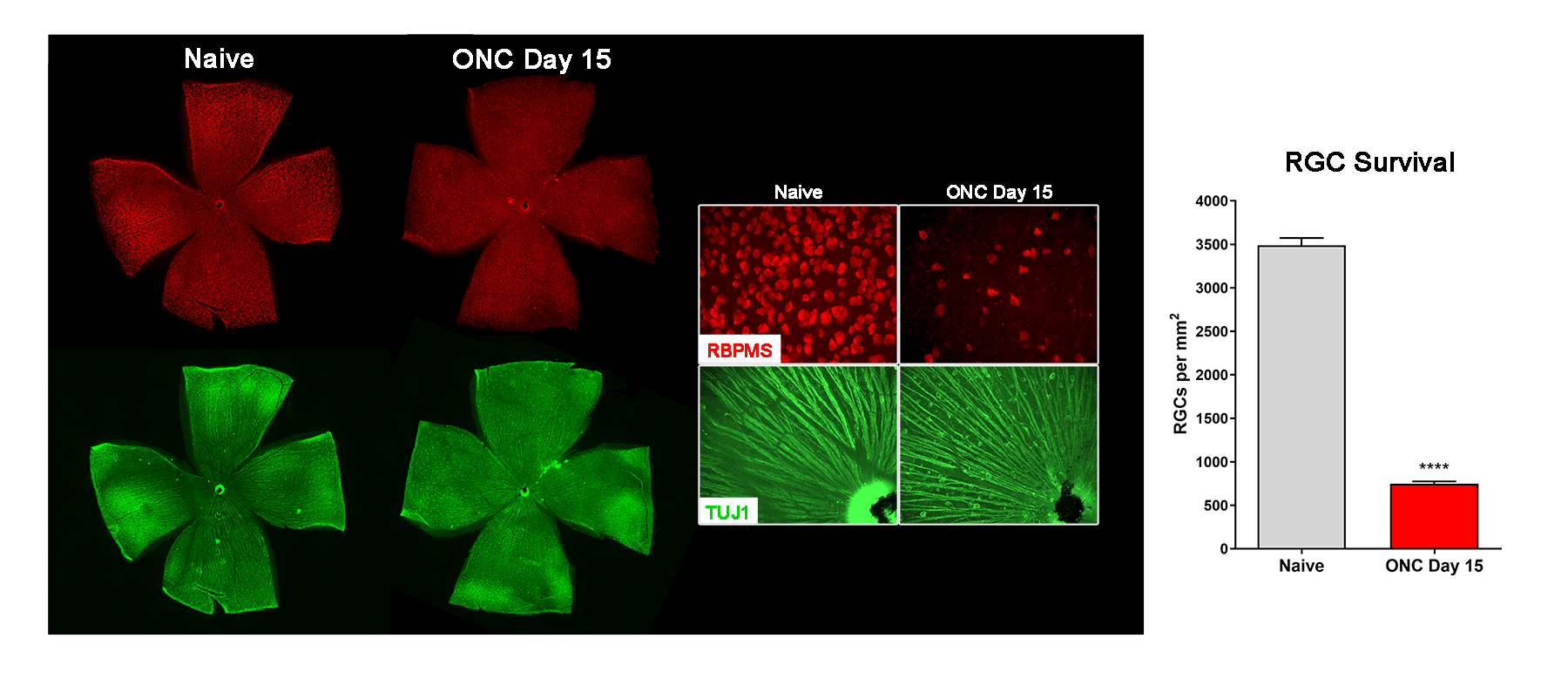
- Retinal flatmounts (RGC survival)
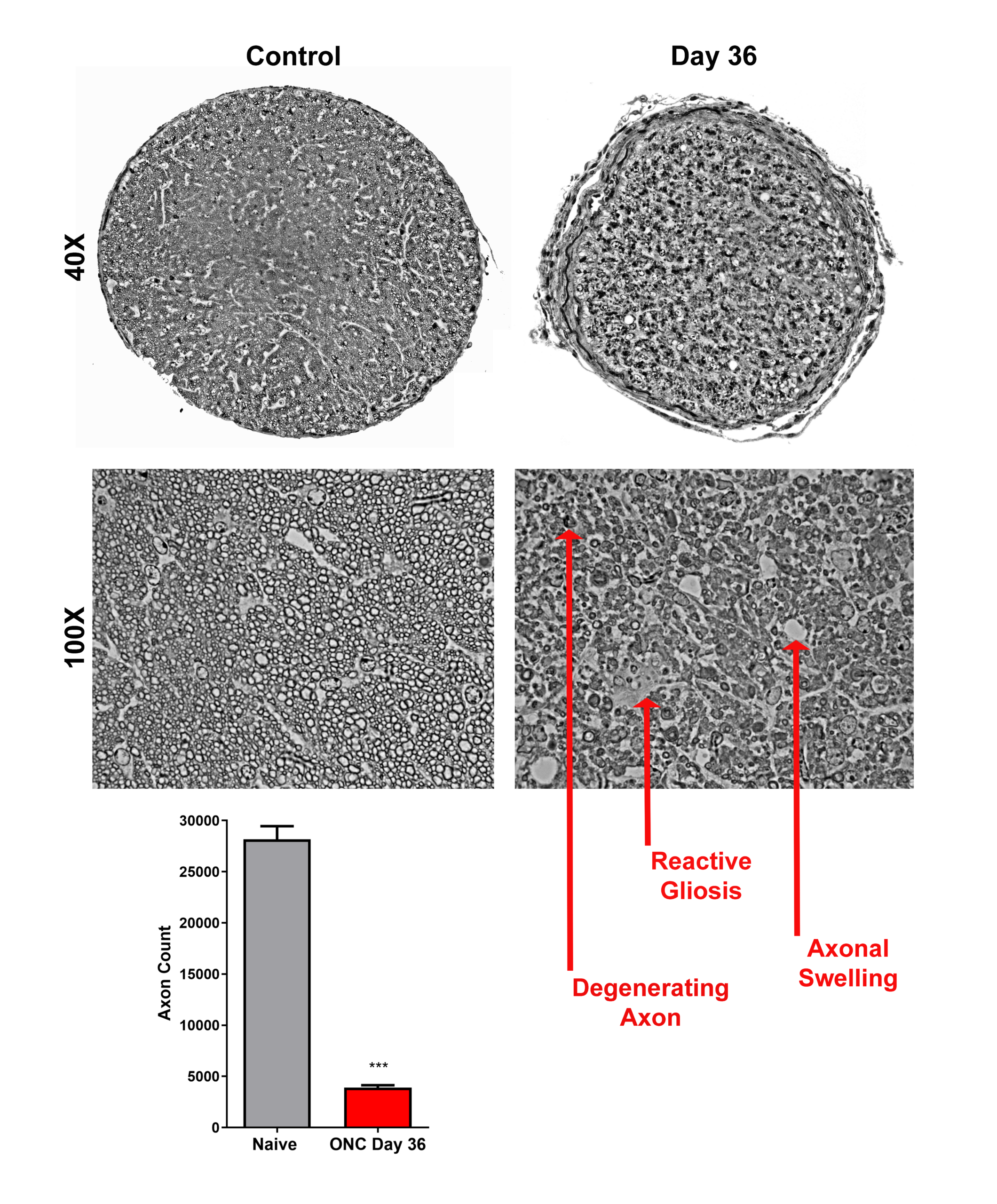
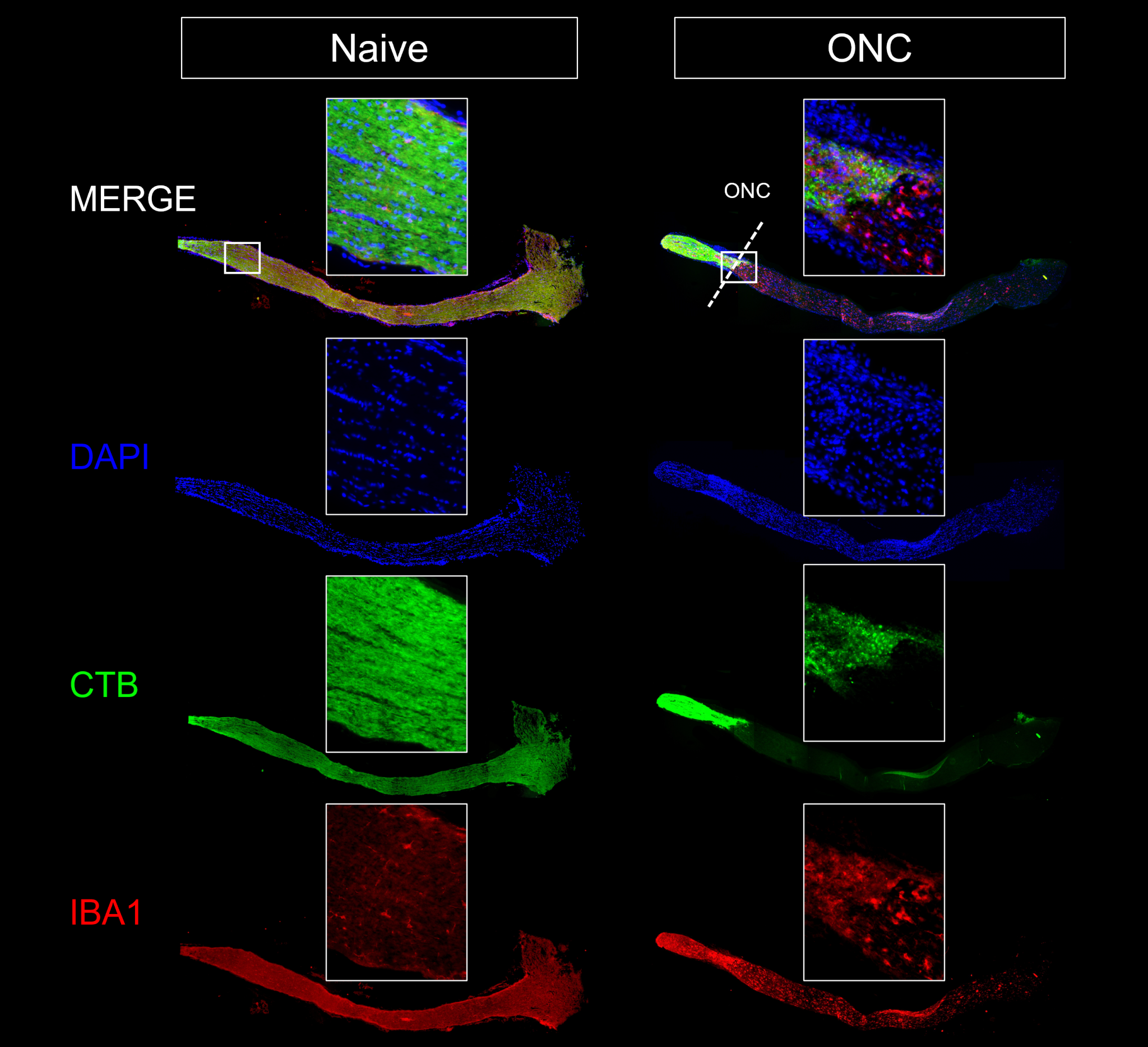
- Optic nerve histology (axon counts, CTB tracing)
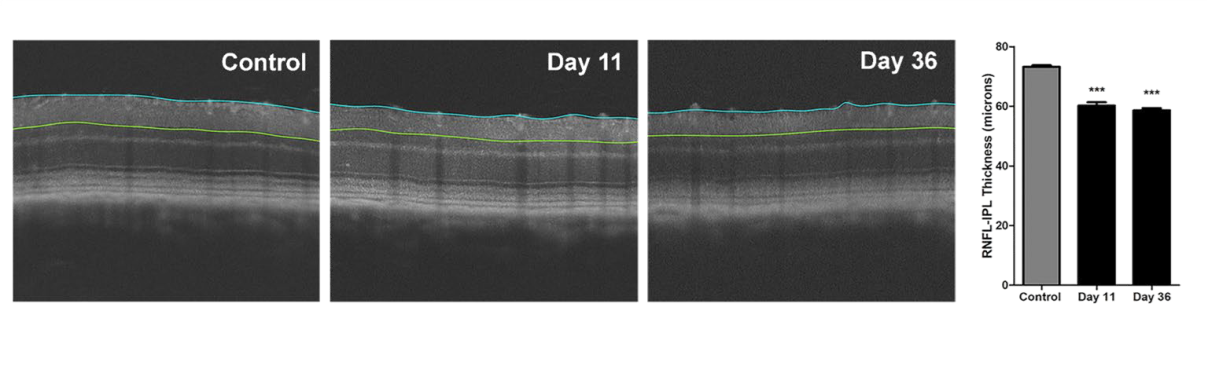
- OCT imaging (ganglion cell complex and total retina thickness)
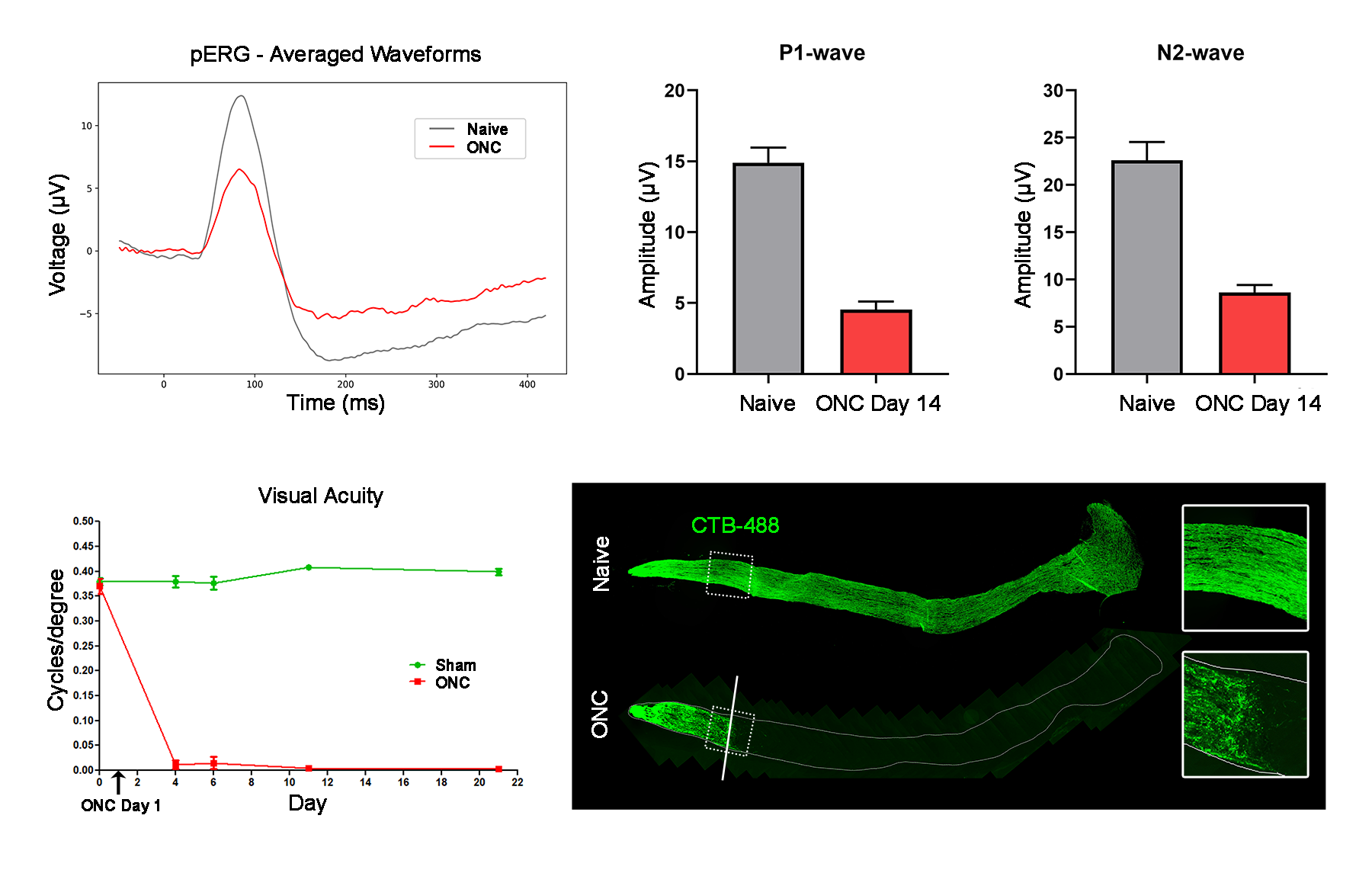
- Pattern ERG (RGC function)
- Optokinetic tracking (visual acuity)
Representative Data
Interested in this model?
We are happy to share more about study design options, endpoints, or timelines.
Contact us to discuss your study goals.

