Oxygen Induced Retinopathy
Pathological retinal neovascularization is a hallmark of several sight-threatening diseases, including retinopathy of prematurity, wet age-related macular degeneration, and proliferative diabetic retinopathy. The oxygen-induced retinopathy (OIR) model is an established system for evaluating therapeutics targeting vessel atrophy, regrowth, pathological neovascularization, and tuft regression.
Model Overview
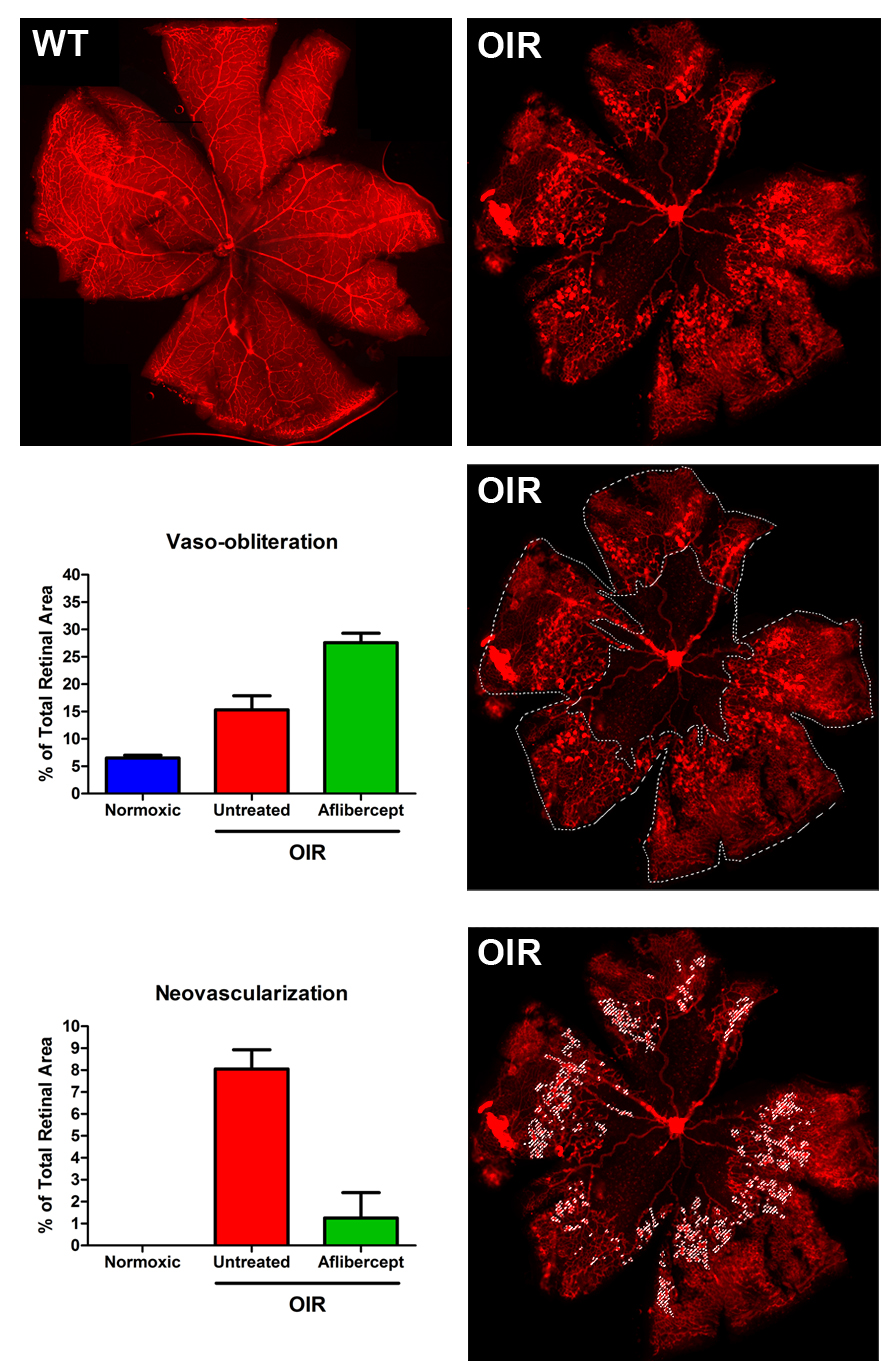
OIR occurs in the developing retina when neonate mouse pups are exposed to high oxygen levels and then returned to a normal oxygen environment. Pups are placed in an O2-regulated chamber set to 75% O2 from postnatal day 8 to 13. Increased oxygen concentrations inhibit production of growth factors critical for vascular development, such as VEGF and erythropoietin, causing vessels in the retina to destabilize and deteriorate, leading to vaso-obliteration around the optic nerve. When pups are returned to room air (21% O2) at P13, the avascular retina becomes hypoxic, activating transcriptional networks governed by hypoxia-inducible factor (HIF) proteins. This generates substantial VEGF expression and initiates a rapid, pathological neovascularization. The neovascular response peaks at postnatal day 18 in the form of “tufts” and regresses between P19 and P26. EyeCRO has standardized and optimized protocols to generate dependable, reproducible, and quantifiable phenotypes.
Typical Endpoints
- Vaso-obliteration quantification on retinal flatmounts
- Neovascular tuft formation quantification on retinal flatmounts
- Vessel regrowth assessment
- Tuft regression assessment
- Fluorescence microscopy and software-based image analysis
Representative Data
Interested in this model?
We are happy to share more about study design options, endpoints, or timelines.
Contact us to discuss your study goals.

