Laser-induced Choroidal Neovascularization
Age-related macular degeneration (AMD) is one of the leading causes of irreversible vision loss in individuals over 50. The neovascular (“wet”) form of AMD is characterized by the growth of abnormal blood vessels from the choroid through Bruch’s membrane into the subretinal space, leading to fluid leakage, hemorrhage, and rapid vision loss. Anti-VEGF therapies have transformed treatment, but there remains significant demand for next-generation therapeutics.
Model Overview
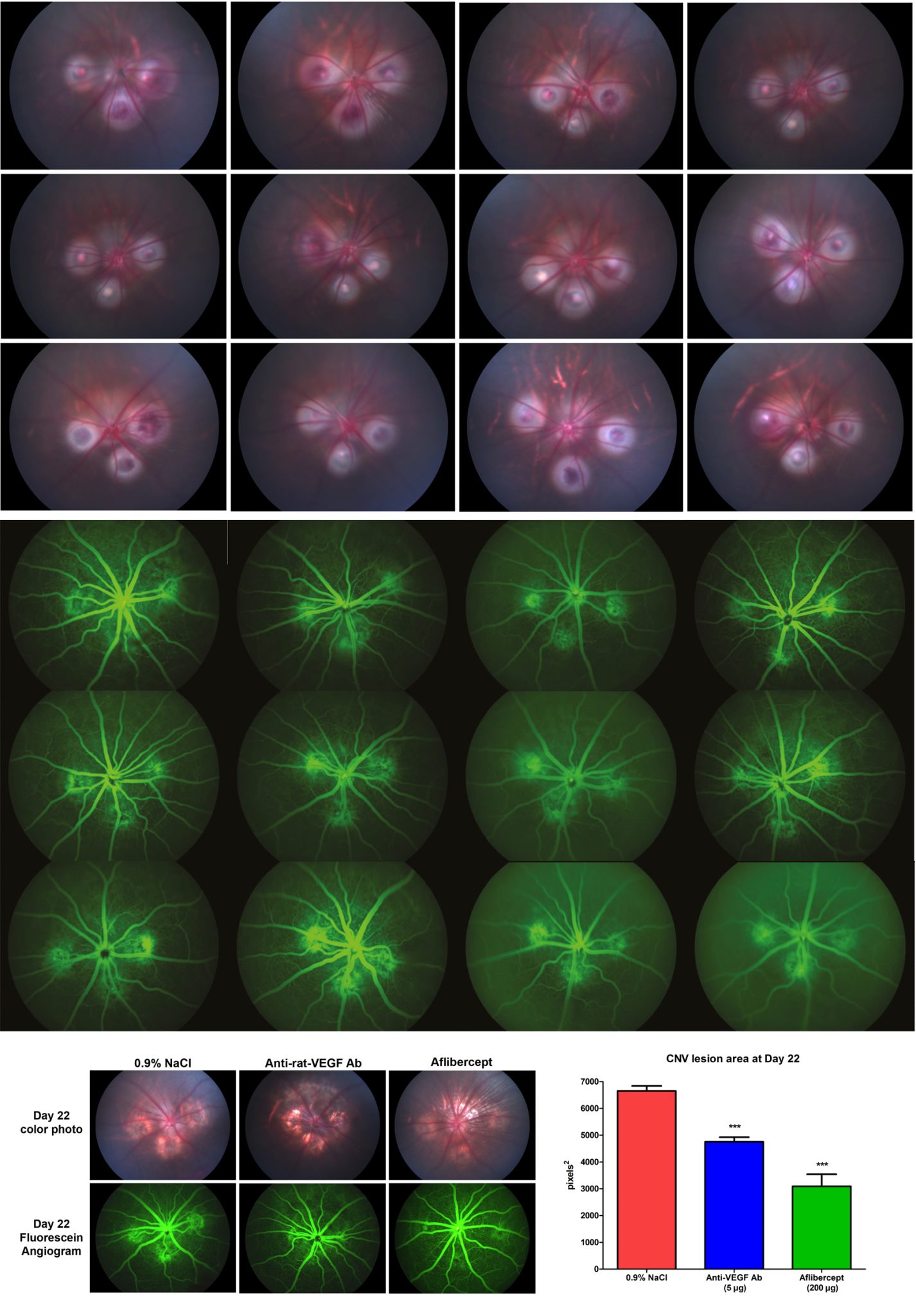
The laser CNV model has been a reliable animal surrogate in the development of therapeutics to treat wet AMD. The pathogenesis of lesion development and regression is conserved among lower mammals, as rodents have been largely predictive of efficacy observed in larger non-human primates and subsequently in human clinical trials. EyeCRO utilizes a Micron III small animal funduscope (Phoenix Research) attached to a thermal laser, which allows fine manipulation of laser focusing and true non-invasive fluorescein angiography without having to sacrifice animals. The same eyes can be used for lesion quantification at multiple time points, and tissue can be harvested at study termination for secondary endpoints such as ELISA, qRT-PCR, immunoblot, LC-MS/MS quantification of drug, or immunohistochemistry.
Typical Endpoints
- Non-invasive fluorescein angiography (lesion leakage quantification)
- Fundus imaging (lesion monitoring at multiple time points)
- OCT imaging (lesion volume and subretinal changes)
- Choroidal/RPE flatmount analysis (lesion area quantification)
- Secondary tissue endpoints: ELISA, qRT-PCR, immunoblot, LC-MS/MS, immunohistochemistry
Representative Data
Interested in this model?
We are happy to share more about study design options, endpoints, or timelines.
Contact us to discuss your study goals.

