Dry Eye Disease
Dry eye disease (DED) is a multifactorial syndrome resulting in dysfunction of the lacrimal functional unit and disruption of normal tear production. It affects up to 33% of the U.S. population, with an estimated societal cost of $55 billion annually. Patients experience symptoms including ocular irritation, redness, decreased tear volume, and reduced quality of life.
Model Overview
DED is an inflammatory autoimmune disease mediated primarily by CD4+ T cells. Antigen-specific Th1 and Th17 cells migrate from regional lymph nodes to the conjunctiva, producing inflammatory cytokines IFN-γ and IL-17A.
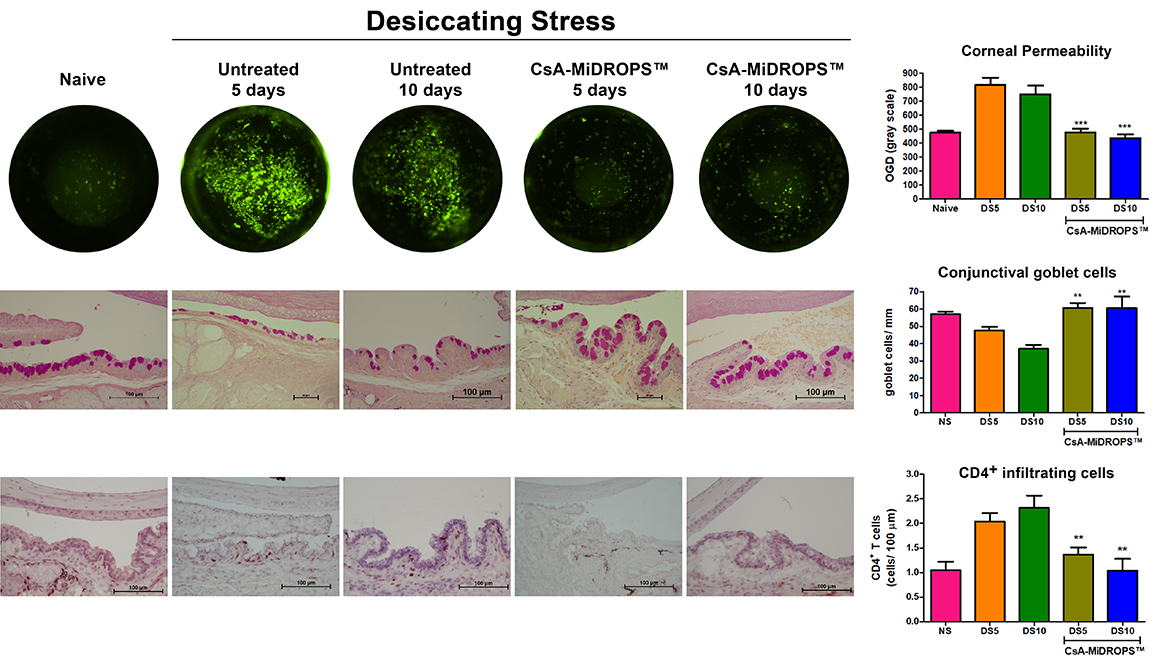
In this model, mice are maintained under strict environmental control of temperature and humidity with constant airflow while receiving daily systemic injections of scopolamine. This induces increased corneal permeability, conjunctival goblet cell loss, CD4+ T cell infiltration, and elevated inflammatory cytokine production, closely recapitulating human DED pathology. EyeCRO can also provide proprietary CsA-MiDROPS® as a positive control for therapeutic efficacy studies.
Typical Endpoints
- Conjunctival goblet cell density
- Quantification of infiltrating CD4+ T cells
- Inflammatory biomarkers: IFN-γ, IL-17A, IL-1β, MMP3, MMP9
- Corneal smoothness evaluation
Representative Data
Interested in this model?
We are happy to share more about study design options, endpoints, or timelines.
Contact us to discuss your study goals.

